To work properly, organic solar cells need more than just a photo-active material absorbing the light and converting it into an electrical current. There are contacts, of course, which extract the current and pass it to the wires. And then there are charge transport layers, which are found in almost all organic solar cells in between the active layer and the contacts.
Ideally, the charge transport layers function as semipermeable membranes. In a solar cell, this means they conduct only one type of carrier (electrons or holes), while blocking the other.
One strategy to achieve that is to use doped organic semiconductors. Chemically, those materials are very similar to the absorber, but with strongly increased conductivity for either electrons (n-doping) or holes (p-doping) due to deliberately introduced impurity molecules, so-called dopants. Using soluble organic materials also for the transport layers would further simplify the device fabrication. Think of three different layers of paint sprayed on top of each other, which would pretty much make a working solar cell.
Sounds great, doesn’t it?
As always, the challenges lie in the practice. One particular issue is that depositing the second and third organic layer must not re-dissolve the underlying ones. This is not trivial given the chemical similarity of the materials used.
In this work, published in ACS Applied Energy Materials, we discuss whether cross-linking the interlayers can help fixing the problem. Cross-linking means to add another ingredient, the cross-linker, which binds the actual functional molecules or polymers together so that they form an insoluble network. Concretely, we tested a novel photo-reactive tris-azide cross-linker, which we incorporated into a p-doped polymer used as a transport layer for holes.
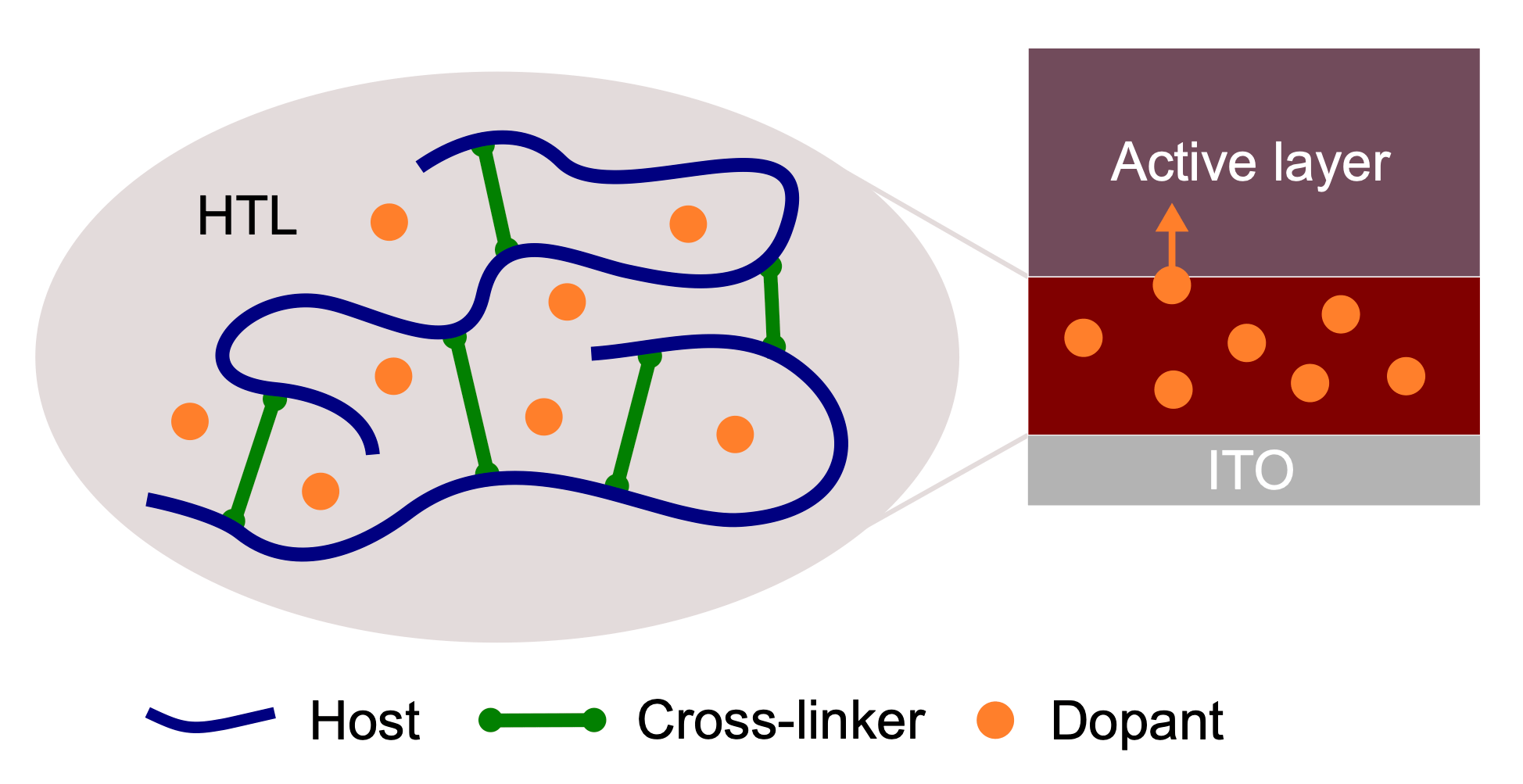
The good news: With our cross-linking strategy, we were able to create a stable, versatile and functional charge transport layer. The less good news, however, is that the solar cells with the cross-linked doped interlayer performed a little worse than control devices with an established standard material.
Using a relatively complex measurement technique called charge extraction by linearly increasing current (CELIV), we found that this is due to unwanted doping of the active layer. Our hypothesis was that these were dopants from the charge transport layer, which were not sufficiently stabilized by the cross-linker and diffused into the active layer. Numerical drift-diffusion simulations showed that such a diffusion would have exactly the observed detrimental effect on the device performance.
Hence, while cross-linking indeed is a promising approach to enable solution-processible charge transport layers, we conclude that cross-linkers must be improved in a way that they not only stabilize the host material but also the dopant.
Reference
Staffan Dahlström, Sebastian Wilken, Yadong Zhang, Christian Ahläng, Stephen Barlow, Mathias Nyman, Seth R. Marder, Ronald Österbacka. Cross-Linking of Doped Organic Semiconductor Interlayers for Organic Solar Cells: Potential and Challenges. ACS Appl. Energy Mater. 4, 14458–14466 (2021)
